Biocides in Food Processing Wastewater
Major Concerns
- Increased TSS in effluent
- Poor settling
- Poor removal of nutrients
Our Solutions
- Restoring nitrification with Vitastim Dynamic Duo
- Introduce new bacterial cultures that grow in difficult conditions
A common concern among the operators of food processing wastewater plants is upsets from something in the influent. Biocides like Quaternary Ammonium compounds (Quats) are a common cleaning component used to disinfect the equipment at the facility. Cleaning crews washing these compounds down the drain and can cause upsets to the treatment plant down stream. These upsets can take the form of poor settling, increased TSS in effluent, or inability to build mixed liquor and subsequent poor removal of BOD and nutrients.
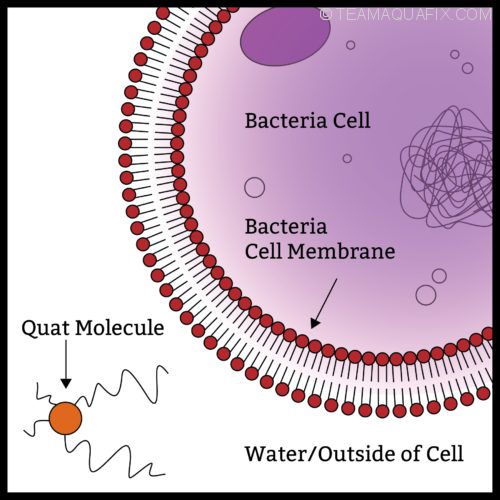
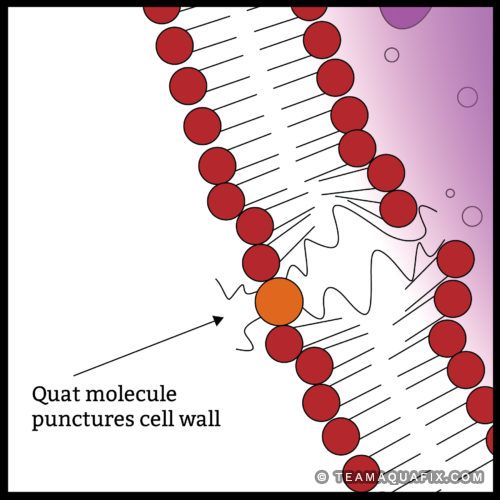
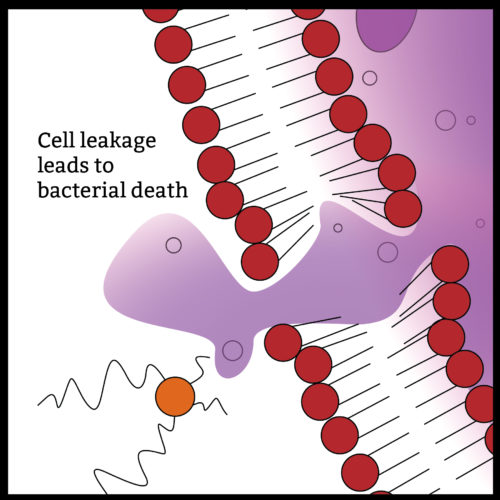
The Quat molecule pictured above in orange works to kill bacteria by first puncturing the outer cell wall. The punctured cell then looses its vital fluids and dies. What is more devastating is that the Quat molecule is then free to act on other cells many times over before it becomes neutralized.
Aquafix is a leader in the research of how biocides and cleaning products affect wastewater treatment plants, and in identifying effective ways to help operators recover their plants after an upset. While the issues can be either acute one time events, or chronic poor performance, the methods for overcoming them rely on similar underlying principals.
Nitrifying bacteria are one of the most fragile bacteria populations in a wastewater plant and the first to be effected when toxicity does hit a treatment plant. Vitastim Dynamic Duo has a proven track record of restoring nitrification when it is lost due to an upset. And a typical treatment only requires 10 days to restore nitrification. When ability to build mixed liquor or poor settling is the issue, we turn to a combination of our Vitsatim Rebuild and SmartBOD. Vitastim Rebuild adds new bacteria cultures to the system that are selected for their ability to floc and grow in difficult conditions. SmartBOD provides the building blocks necessary to accelerate the maturation of the bacteria population. In many cases a basic microanalysis by our microbiologists can help pinpoint the greatest areas of need in the bacterial population, and help us to tailor a treatment specific to the unique needs of the system.
Let our expert team of scientists and researchers in the Aquafix Laboratory help you solve all your food processing biocide issues.
Looking for more solutions and information? We have so much more to share! Watch our upcoming webinars page for topics related to your needs. Or contact us directly at 888.757.9577 and info@teamaquafix.com
